Clenbuterol is a bronchodilator used in horses for the management of lower airway disease, including equine asthma and other conditions associated with bronchoconstriction. These disorders occur when the small airways narrow due to inflammation, excess mucus, and smooth muscle contraction, leading to coughing, nasal discharge, and exercise intolerance.
Classified as a beta-2-adrenergic agonist, clenbuterol works by stimulating beta-2 receptors in bronchial smooth muscle. This activation causes relaxation of the airway muscles, resulting in dilation of the bronchi (lower airways) and improved airflow. In addition to its bronchodilatory effects, clenbuterol may enhance mucociliary clearance, helping remove mucus from the respiratory tract.
Despite its therapeutic benefits, clenbuterol must be used carefully. Prolonged or excessive dosing can lead to receptor downregulation, reducing its effectiveness over time. Because it stimulates the sympathetic nervous system, potential side effects include sweating, muscle tremors, tachycardia (rapid heart rate), and excitability.
Clenbuterol is typically prescribed as part of a broader management strategy that includes environmental modification and control of airway inflammation. When used appropriately under veterinary guidance, it can improve respiratory comfort and performance in affected horses.
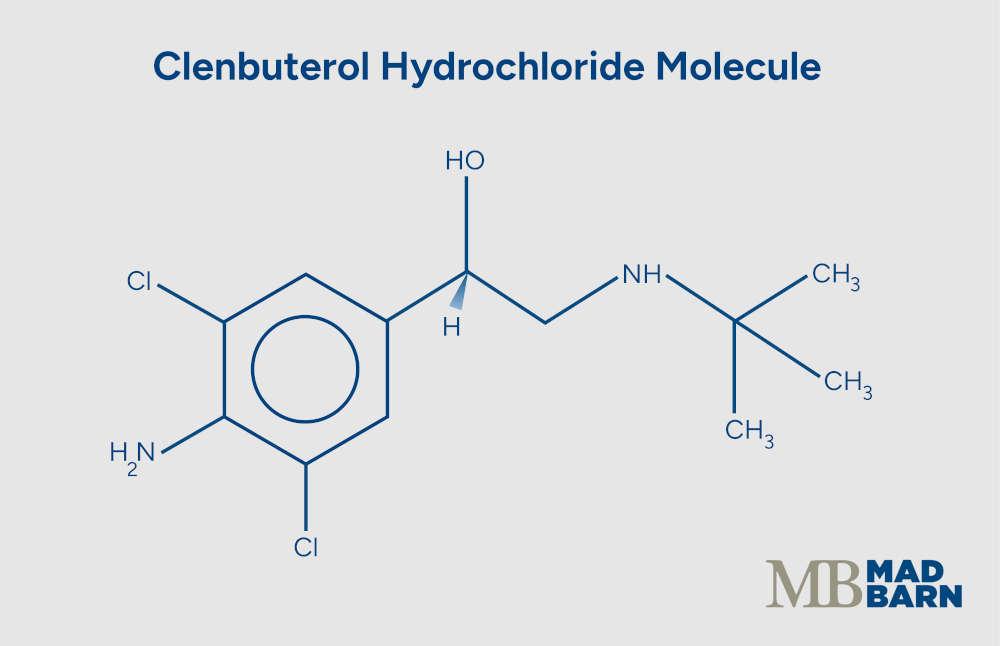
Clenbuterol Hydrochloride for Horses
Clenbuterol is a bronchodilator and mucokinetic drug commonly prescribed to horses as treatment for constriction of the lower airways (the bronchi). [1][2] Bronchodilators relax the smooth muscles of the airway and ease clearance of mucus, improving airflow and reducing the effort required to breathe.
Narrowing or obstruction of the bronchi (bronchospasm) is a symptom of various equine respiratory diseases including severe equine asthma, sometimes called recurrent airway obstruction (RAO) or heaves. [1] A veterinarian may prescribe clenbuterol in combination with a corticosteroid to treat the underlying inflammatory condition that causes bronchospasm. [1][2]
Clenbuterol is a long-acting beta-2 adrenergic agonist. It relaxes airway smooth muscle, supports mucociliary clearance, and at high or sustained doses, it can affect body composition in horses. [1][2][3]
Clenbuterol is the only beta-2 agonist currently approved by the U.S. FDA for oral use in horses. [3] Equestrian sport regulatory bodies prohibit it in competition, so withdrawal times apply. [4]
Available Forms
Clenbuterol hydrochloride is available for horses in oral and intravenous forms. [5] The oral formulation is the only product approved for use in horses and is marketed under the trade name Ventipulmin® Syrup.
The injectable formulation is marketed under the trade name Planipart® and is primarily labeled to relax uterine smooth muscle in pregnant cattle. Its use differs from the oral syrup formulation, which is intended for respiratory conditions. [5]
Clenbuterol is commonly prescribed alongside a corticosteroid, such as dexamethasone or prednisolone, when treating inflammatory airway disease. While clenbuterol provides bronchodilation by relaxing airway smooth muscle, corticosteroids address the underlying inflammation. Combination therapy is frequently used in horses with moderate to severe airway disease. [1][2]
What's your top priority with your horse's health?
Drug Class: Beta-2 Adrenergic Agonists
Beta-2 adrenergic agonists are a class of drugs that stimulate beta-2 adrenergic receptors, which are part of the sympathetic nervous system. [6]
These receptors are found primarily in:
- Bronchial smooth muscle (lungs)
- Uterine smooth muscle
- Vascular smooth muscle
- Skeletal muscle
Beta-2 adrenergic agonists are used to treat respiratory disorders involving hyper-contractility of the lower respiratory tract (the bronchi). They are usually prescribed for concurrent use with corticosteroids. [6] Common disorders treated with beta-2 adrenergic agonists include chronic obstructive pulmonary disease (COPD) and asthma. [1][6]
Other names for clenbuterol include: [5]
- Bronq-C
- NAB-365
- Clebuterolum
- Bronchopulmin®
- Dilaterol®
- Respipulmin®
- Broncodil®
- Clenasma®
- Cesbron®
- Clenbutol
- Monores®
- Prontovent®
- Spiropent®
Other beta-2-adrenergic agonists used in horses include: [1]
- Albuterol
- Salbutamol
- Pirbuterol
- Fenoterol
Mechanism of Action
Beta-2 adrenergic agonists work by activating beta-2 adrenergic receptors, which are normally activated by epinephrine (adrenaline) or norepinephrine. During the “flight or fight” response, several types of alpha and beta adrenoceptors are activated. This increases heart rate and relaxes smooth muscle, which results in increased blood flow and vasodilation (widening of the blood vessels). [1]
Clenbuterol works by stimulating beta-2 receptors on smooth muscle cells. When these receptors are activated, they trigger a chain reaction inside the cell that increases levels of a messenger molecule called cyclic AMP (cAMP). [7]
Higher cAMP levels signal the airway muscles to relax. Specifically, clenbuterol reduces the activity of enzymes that promote muscle contraction and alters ion movement across the cell membrane. In the bronchi (lower airways), this leads to relaxation of smooth muscle and widening of the airways (bronchodilation). [7]
Clenbuterol also helps improve the airway’s ability to clear mucus. By stimulating beta-2 receptors, it increases the movement of tiny hair-like structures (cilia) that line the respiratory tract. These cilia sweep mucus and trapped debris up and out of the airways, a process known as mucociliary clearance. [8][9]
At the same time, clenbuterol reduces mucus production from airway glands. Together, these effects help keep the airways clearer and may improve breathing in horses with inflammatory airway disease. [8][9]
Uses in Horses
In horses, clenbuterol is only approved for management of airway obstruction due to bronchospasm and mucus accumulation. [1][5] The primary indication is recurrent airway obstruction (RAO) or heaves, such as from chronic obstructive pulmonary disease or asthma. It may also provide relief during recovery from pneumonia or smoke inhalation. [1]
It’s important to note that bronchodilators do not cure disease, they only provide symptomatic relief. Treatment of COPD or asthma typically requires reducing dust exposure and pairing clenbuterol with a corticosteroid such as dexamethasone. [1]
Off-Label Uses
Clenbuterol is only approved for management of airway obstruction in horses. [5] However, veterinarians may prescribe it for other conditions based on existing research, which is referred to as off-label use.
Medications are prescribed off-label at the discretion of the treating veterinarian, and the drug manufacturers do not guarantee these applications are safe. Horse owners should not administer drugs off-label without direct instructions from a licensed veterinarian.
Off-label uses of clenbuterol in horses include: [1][2][5]
- Emergency use in high-risk pregnancy or dystocia: By relaxing smooth muscle in the uterus, injectable clenbuterol can be used to delay the onset of labor or reduce uterine contraction strength to allow manipulation of the fetus.
- Repartitioning: Research has demonstrated changes in body composition at higher doses; however, such use is not medically indicated in horses and carries significant risk.
Off-label use of clenbuterol in animals intended for human consumption is prohibited by law. [5]
Administration Routes
In the United States, Ventipulmin® syrup is the only form of clenbuterol available for horses and is given orally. In Europe, Australia, Canada, and South America, Ventipulmin® has been available in multiple forms (e.g., syrup, gel, injectable) for many years. [1]
The U.S. label recommends a variable dose schedule. The FDA required proof of efficacy in the absence of other treatments, so the U.S. label allows incremental dosing; the maximum dose on the label must be approached gradually to avoid side effects (sweating, trembling, tachycardia, excitement). [1]
The injectable form Planipart® is sometimes used off-label in horses, and is administered intravenously by a veterinarian or licensed veterinary technician.
Dosage
The oral formulation of clenbuterol is prescribed initially at a dosage of 0.8 μg/kg body weight, administered twice daily. Depending on the horse’s response to treatment, the prescribing veterinarian may increase the dosage every three days to a maximum dosage of 3.2 µg/kg body weight administered twice daily. [5]
If the horse does not respond favorably after 30 days of treatment, clenbuterol is discontinued. [5]
Storage
Store clenbuterol syrup at or below 25°C (77°F); avoid freezing. Keep in a closed container. Clenbuterol injection should be stored below 25°C (77°F) and discarded 28 days after first use. [5]
Always follow manufacturer label for the specific formulation.
Safety Warnings & Precautions
As with all medications, clenbuterol should be used at the lowest effective dose for the shortest duration necessary. [1][2][5]
Higher or prolonged dosing has been associated with: [1][2][5]
- Reduced aerobic capacity
- Altered cardiac function
- Increased risk of adverse events
Horses with cardiac disease should not receive clenbuterol. [5]
Monitoring
During treatment, monitor for signs of excessive beta-adrenergic stimulation, including: [5]
- Tachycardia (rapid heart rate)
- Excitement
- Sweating
- Muscle tremors
If signs of beta-adrenergic or other side effects are noticed, consult the prescribing veterinarian. Do not make changes to your horse’s dosing schedule without veterinary guidance.
For horses on longer courses, periodic assessment of cardiac and respiratory function may be recommended. Chronic use at doses above the minimum should be avoided. [1][2]
Contraindications
Contraindications refer to circumstances where a drug is to be avoided or only used with caution.
Clenbuterol should be used with caution for: [7][12]
- Horses with cardiac disease: Potential for tachycardia and increased cardiac workload
- Breeding stallions and broodmares: Clenbuterol may diminish normal uterine contractility, induce abortion and reduce fertility
- Near-term mares: Clenbuterol relaxes uterine muscle and can delay onset of labor or complicate delivery
Side Effects
Beta-2 agonists like clenbuterol are selective for beta-2-adrenoceptors, but at high enough doses will activate beta-1-adrenoceptors, which can lead to side effects. [1]
Activation of beta-1-adrenoceptors can cause: [1]
- Tachycardia (rapid heart rate)
- Excitement and restlessness
- Sweating
- Trembling
- Muscle tremors
- Urticaria (hives)
Long-term use of clenbuterol in horses has been associated with: [2][5]
- Reduced aerobic capacity
- Tachyphylaxis (rapid, severe decline in drug effectiveness)
- Reduced endocrine, immune and reproductive function
Chronic administration at higher therapeutic doses has been associated with negative effects on aerobic performance, cardiac function, and recovery from exercise in horses. [2][10] Long-term beta-2-agonist use in other species has been associated with cardiac hypertrophy and, in some cases, myocardial damage. [2][10]
Compounded clenbuterol products have been associated with dosing errors and toxicity. Whenever possible, FDA- or Health Canada–approved formulations should be used. [12]
Acute Toxicity (Overdose)
Signs of overdose are consistent with beta-1-adrenoceptor stimulation and include: [1]
- Sweating
- Trembling
- Tachycardia (rapid heart rate)
- Excitement
If a horse shows signs of overdose, move them to a quiet stall and contact your veterinarian right away. [1]
Depending on when the dose was given, gastrointestinal decontamination may be appropriate. [11]
Management of toxicity may include administration of: [11]
- Fluids and electrolytes
- Insulin
- Parenteral beta blockers to control heart rate and blood pressure
- Benzodiazepines to manage agitation or seizures

Drug Interactions
Formal drug interaction studies for clenbuterol in the horse are limited. Because clenbuterol is a beta-2-agonist, use with beta-blockers such as propranolol or sotalol may antagonize clenbuterol’s effects. [5]
Drugs that may enhance the side effects of clenbuterol include: [5]
- Sympathomimetics
- Theophylline/aminophylline
- Tricyclic antidepressants
Drugs that may increase risk of cardiac arrhythmias when administered with clenbuterol include: [5][13]
- Inhalant anesthetics
- Digoxin
Clenbuterol may antagonize (reduce) the effects of the following drugs: [5]
- Dinoprost
- Oxytocin
- Xylazine and other alpha-2 agonists
Corticosteroids (e.g., dexamethasone) are commonly used alongside clenbuterol for airway disease. Low doses of clenbuterol combined with dexamethasone are considered beneficial for managing airway inflammation. [2]
Regulatory Status & Legal Considerations
Clenbuterol is approved in both Canada and the United States for use in horses with airway obstruction associated with inflammatory airway disease, including recurrent airway obstruction (RAO). [1]
In the U.S., it is marketed as Ventipulmin® Syrup and is specifically labeled for equine use.
Veterinarians prescribe the oral formulation to horses according to manufacturer guidelines. As with all prescription medications, it must be used under veterinary supervision to ensure appropriate dosing and monitoring.
In some cases, veterinarians may use clenbuterol in a manner that differs from the product label. Such use falls under established veterinary prescribing regulations and relies on clinical judgment, published research, and experience with the drug in horses.
Because clenbuterol is also known to be misused in other species for growth-promoting effects, its distribution and use are strictly regulated in many jurisdictions. [14] In horses, it should only be used for legitimate medical indications as prescribed by a veterinarian.
If your veterinarian recommends clenbuterol, it is appropriate to discuss expected benefits, duration of therapy, potential side effects, and any competition withdrawal requirements that may apply.
Status in Competition
Clenbuterol is classified as a performance-affecting medication in many competitive disciplines.
Clenbuterol is listed on the FEI Prohibited Substances List as a banned substance in competition and is not permitted in horses at the time of competition. [4][15][16] Positive tests in horses may result in sanctions, including disqualification and suspension.
Because clenbuterol is also prohibited under human anti-doping regulations, riders may face disciplinary action if it is detected in their own samples. [16]
At the minimum dosage of 0.8 µg/kg given twice daily, the withdrawal time is listed as 168 hours (7 days). The product may remain detectable in urine samples for longer, with a withdrawal time of approximately 288 hours (12 days). [7]
Always check the regulations for your specific sport before starting a new medication for your horse.
Frequently Asked Questions
Here are some frequently asked questions about clenbuterol use in horses:
Clenbuterol is used to manage airway obstruction in horses with respiratory conditions such as equine asthma and recurrent airway obstruction (heaves). It relaxes smooth muscle in the airways to improve airflow and supports mucociliary clearance.
Clenbuterol is a beta-2 adrenergic agonist. It binds to beta-2 receptors in the airways, which results in relaxation of smooth muscle leading to bronchodilation (widening of the airways). It may also increase ciliary beat frequency, which can support mucociliary clearance.
Clenbuterol is administered to horses orally twice a day. The prescribing veterinarian may increase the dosage every three days, depending on the horse's response to treatment. If the horse does not respond to treatment within 30 days, clenbuterol is discontinued. It is usually prescribed concurrently with corticosteroids. Less commonly, veterinarians may administer intravenous clenbuterol off-label to pregnant mares.
Potential side effects include increased heart rate, excitement or restlessness, sweating, and muscle trembling, especially if the dose is too high or increased too quickly. Clenbuterol should be used with caution in horses with cardiovascular disease, and veterinary guidance is important to reduce the risk of adverse events.
Clenbuterol is generally used for short-term relief of bronchospasm rather than as a long-term standalone strategy. Repeated or prolonged use may be less effective over time and may increase the risk of side effects. Long-term management of equine asthma typically emphasizes environmental control and anti-inflammatory therapy, with bronchodilators used as needed under veterinary direction.
Studies have not shown a meaningful improvement in aerobic performance in healthy horses at therapeutic doses. Bronchodilation in normal horses is typically small and is not associated with improved oxygen consumption. Use for performance enhancement is prohibited in competition.
The FEI lists clenbuterol as a performance enhancing drug and both horse and rider are prohibited from use during competition. The withdrawal time for the minimum dosage is ~7 days. Always check the regulations for your sport before starting medication for your horse.
Withdrawal guidance varies by governing body, dosing protocol, and testing approach. The FEI lists a withdrawal time of 168 hours (about 7 days) at a dosage of 0.8 micrograms per kilogram twice daily, and urine may test positive longer (about 288 hours). Always consult current competition regulations and your veterinarian before administering clenbuterol close to an event.
Summary
Clenbuterol is a beta-2-adrenergic agonist bronchodilator and mucokinetic drug used in horses to manage airway obstruction associated with respiratory conditions such as equine asthma and heaves.
- Clenbuterol provides symptomatic relief of bronchospasm and mucus accumulation
- It is only available by prescription and should be administered according to the directions from the treating veterinarian
- It works by relaxing airway smooth muscle and increasing mucociliary clearance
- Clenbuterol is usually prescribed concurrently with corticosteroids and is only used for short-term symptom management
- Side effects can include tachycardia, excitement, sweating, and tremors
- It is prohibited in competition, withdrawal time for the minimum dosage is ~7 days
References
- Robinson. N. E. Clenbuterol and the Horse. AAEP Proceedings. 2000.
- Kearns. C. F. and McKeever. K. H. Clenbuterol and the Horse Revisited. The Veterinary Journal. 2009. View Summary
- Chuang. M. S. et al. Detection of Urine and Blood Clenbuterol Following Short-Term Oral Administration in the Horse. Immunopharmacology and Immunotoxicology. 2010. View Summary
- FEI Clean Sport. FEI. 2025.
- Plumb. D. C. Plumb's Veterinary Drug Handbook: Desk. John Wiley & Sons. 2018.
- Cazzola. M. et al. beta-2-Adrenoceptor Agonists: Current and Future Direction. British Journal of Pharmacology. 2011.
- Soma. L. R. et al. Pharmacokinetics and Disposition of Clenbuterol in the Horse. Journal of Veterinary Pharmacology and Therapeutics. 2004. View Summary
- Norton. J. L. et al. Effect of Clenbuterol on Tracheal Mucociliary Transport in Horses Undergoing Simulated Long-Distance Transportation. Journal of Veterinary Internal Medicine. 2013. View Summary
- Laan. T. T. J. M. et al. The Effect of Aerosolized and Intravenously Administered Clenbuterol and Aerosolized Fluticasone Propionate on Horses Challenged with Aspergillus Fumigatus Antigen. Veterinary Research Communications. 2006. View Summary
- Sleeper. M. M. et al. Chronic Clenbuterol Administration Negatively Alters Cardiac Function. View Summary
- Thompson. J. A. et al. Effects of Clenbuterol Administration on Serum Biochemical, Histologic, and Echocardiographic Measurements of Muscle Injury in Exercising Horses. American Journal of Veterinary Research. American Veterinary Medical Association. 2012. View Summary
- Thompson. J. A. et al. Clenbuterol Toxicosis in Three Quarter Horse Racehorses after Administration of a Compounded Product. Journal of the American Veterinary Medical Association. 2011. View Summary
- Stegmann. G. F. and Bester. L. Some Cardiopulmonary Effects of Midazolam Premedication in Clenbuterol-Treated Bitches during Surgical Endoscopic Examination of the Uterus and Ovariohysterectomy. Journal of the South African Veterinary Association. 2001.
- Davis. J. L. et al. Update on Drugs Prohibited from Extralabel Use in Food Animals. Journal of the American Veterinary Medical Association. 2009.
- FEI Tribunal Issues Final Decision in Human Doping Case. FEI. 2024.
- The Prohibited List. World Anti Doping Agency.