Methocarbamol is a centrally acting muscle relaxant commonly used in horses to treat skeletal muscle spasms. These spasms can occur during conditions that cause muscle inflammation or injury and are often associated with disorders such as exertional rhabdomyolysis, also known as “tying-up.”
Methocarbamol works primarily by lowering activity in the central nervous system (CNS). Rather than acting directly on muscle tissue, the drug reduces abnormal nerve signals in the brain and spinal cord that trigger persistent muscle contractions.
By interrupting this cycle of pain and muscle spasm, methocarbamol helps affected muscles relax while generally preserving muscle strength and voluntary movement.
Methocarbamol is frequently used as part of a broader treatment plan for horses experiencing acute muscle injury, tying-up episodes, or certain neurological conditions that cause muscle rigidity. As a regulated medication in many equestrian disciplines, appropriate withdrawal times and veterinary guidance are essential when treating performance horses.
Methocarbamol (Robaxin®) for Horses
Methocarbamol is a centrally acting skeletal muscle relaxant utilized in both human and veterinary medicine to alleviate the discomfort associated with acute, painful musculoskeletal conditions and to reduce severe muscular spasms.
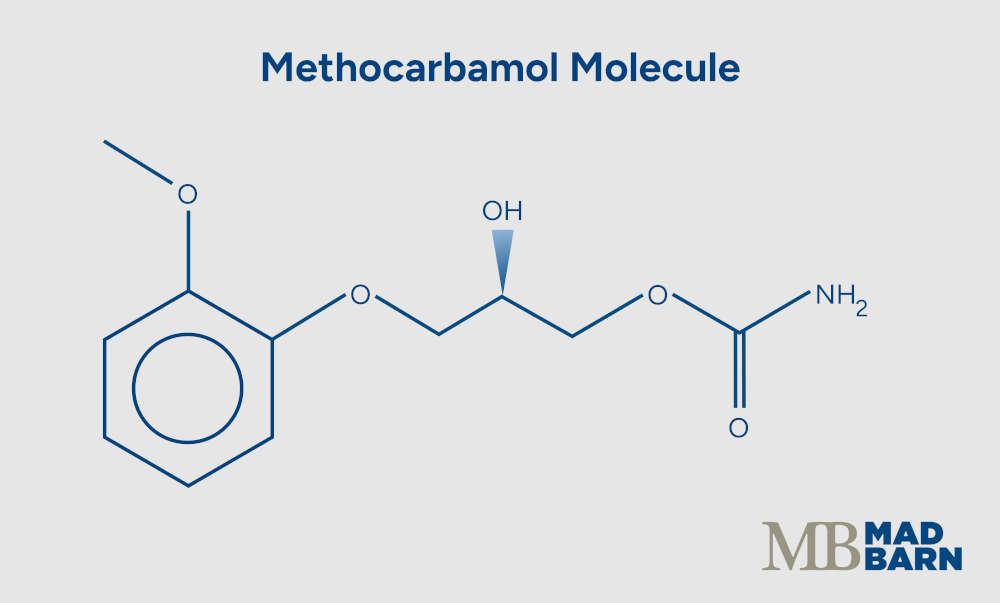
The compound is approved by the FDA for human and veterinary use and is chemically classified as a carbamate derivative of guaifenesin. [1]
In equine medicine, methocarbamol is frequently used as part of a broader treatment plan that includes:
- Stall rest
- Physical rehabilitation
- Non-steroidal anti-inflammatory drugs (NSAIDs)
The drug functions primarily as a central nervous system (CNS) depressant. Unlike neuromuscular blocking agents used during general anesthesia, methocarbamol does not disrupt normal neuromuscular transmission or directly affect muscle contractions. [2][3]
Instead, it exerts selective action on the internuncial neurons of the spinal cord, leading to a reduction in skeletal muscle hyperactivity without a loss of normal muscle tone. [2]
Available forms
Methocarbamol is commercially available in several formulations, though the regulatory approval status of these forms varies significantly within equine medicine.
The most well-known brand name in veterinary medicine is Robaxin®-V.
Injectable Solutions
The parenteral formulation of methocarbamol is FDA-approved for intravenous (IV) administration in horses. Because methocarbamol is sparingly soluble in water, the injectable solution is formulated using polyethylene glycol as a co-solvent. [2][4]
Oral Tablets
Tableted formulations are available at various dosages. While these tablets are FDA-approved for use in dogs, cats, and humans, there is no FDA-approved oral tablet specifically labeled for equine use. Given this, prescription of oral methocarbamol tablets to horses is considered off-label. [5][6]
Due to the large volume of tablets required for a therapeutic dose in a mature horse, tablets are typically crushed, suspended in water, and administered via a nasogastric tube or oral dose syringe.
Compounded Oral Formulations
Veterinary compounding pharmacies frequently formulate methocarbamol into oral pastes, flavored suspensions, and bulk powders specifically concentrated for equine use.
These compounded formulations are also classified as off-label applications, but offer improved ease of administration for horse owners.
What's your top priority with your horse's health?
Drug Class: Skeletal Muscle Relaxants
Skeletal muscle relaxants are a broad class of drugs used in veterinary and human medicine to alleviate muscle spasms, spasticity, and associated pain. This category is functionally divided into two distinct groups based on their primary site of action: centrally acting agents and direct-acting agents.
Centrally acting muscle relaxants work within the central nervous system, affecting nerve cells in the brain and spinal cord known as interneurons. [2]
Normally, these nerve pathways help control reflexes that cause muscles to contract. When these pathways become overstimulated due to injury or disease, they can trigger repeated or sustained muscle contractions.
Methocarbamol works by reducing nerve signals along these pathways, which helps relax the muscles and decrease involuntary tension.
This mechanism differs from direct-acting muscle relaxants, such as dantrolene. Instead of acting on the nervous system, dantrolene works directly on the muscle fibers. It prevents the release of calcium from inside muscle cells, which disrupts the process that causes muscles to contract. [3]
Other skeletal muscle relaxant drugs used in horses include: [3]
- Guaifenesin
- Diazepam
- Dantrolene
Mechanism of Action
The exact pharmacological mechanism of methocarbamol is not fully understood. However, research confirms that it acts on the central nervous system rather than directly on the muscle tissue or peripheral nerves. [3]
Methocarbamol acts selectively on the internuncial neurons (interneurons) of the spinal cord and the midbrain reticular activating system. Interneurons act as critical relays between sensory (afferent) and motor (efferent) neurons. [2][7]
During instances of acute muscle trauma, excessive pain signaling causes hyperexcitability along interneuron pathways, resulting in sustained, involuntary muscle contraction. Methocarbamol produces a blockade against the action of these reflexes, effectively decreasing nerve transmission. [2][7]
Electrophysiological studies indicate that methocarbamol increases the refractory period of muscle cells, meaning it takes longer for the muscle to reset between contractions. However, methocarbamol has no direct relaxant effect on the contraction apparatus of striated muscle. [4]
Following administration, the compound undergoes significant metabolism in the liver. [3][7]
Uses in Horses
Methocarbamol is utilized in equine veterinary medicine for a variety of conditions affecting the musculoskeletal and nervous systems.
The injectable form is approved as an adjunct therapy for treatment of acute inflammatory and traumatic conditions of the skeletal muscle. It is administered to reduce muscle spasms and induce relaxation in striated muscle tissue. [4]
Intravenous administration of methocarbamol in horses is commonly used to address muscle spasms in cases of:
- Trauma and sprains: Muscular and ligamentous sprains, strains, and generalized blunt force trauma causing secondary muscle hyper-contraction
- Inflammatory myopathies: Where localized inflammation triggers reflex muscle guarding, such as in cases of myositis, fibrositis, bursitis, and synovitis
- Rhabdomyolysis (tying-up): a painful condition characterized by profound muscle cramping, stiffness, and life-threatening kidney damage, often triggered by exercise
- Tetanus: Employed to maintain muscle relaxation and counteract the severe, life-threatening tetanic spasms induced by the neurotoxin tetanospasmin
- Surgical adjunct: To alleviate muscular spasm prior to or following surgical procedures
Off-Label Uses
“Off-label” (or extra-label) use refers to administration of a drug in a manner that is not explicitly described on the FDA-approved package insert. This includes using the drug in a non-approved species, for a non-approved medical condition, or via a non-approved route of administration.
Off-label use is permitted under veterinary prescribing regulations when no approved equine alternative is available. In these cases, veterinarians rely on published research, clinical experience, and established treatment protocols to determine appropriate dosing and monitoring.
In horses, oral administration of methocarbamol is strictly off-label, as there is no FDA-approved oral methocarbamol product specifically labeled for equines. [5][6]
Despite this, oral methocarbamol is often used by equine practitioners for ongoing management of the conditions listed above. It’s particularly common in the management of chronic back and neck pain, generalized stiffness in performance horses, and ongoing rehabilitation from orthopedic injuries.
Administration Routes
The administration of methocarbamol in horses requires precise calculation based on the animal’s exact body weight, the severity of the muscle pathology, and the selected route of administration. The intravenous formulation must be administered by a licensed veterinarian or veterinary technician.
Intravenous (IV) Administration
The parenteral route is used for acute, severe clinical presentations requiring immediate therapeutic intervention (e.g., severe exertional rhabdomyolysis or tetanus).
The injectable solution requires precise injection directly into the jugular vein, which is why only qualified veterinary personnel can administer the intravenous form. [4]
Rapid injection is generally recommended to achieve the most effective immediate response, but extreme care must be taken to ensure the needle remains strictly intravascular. [4]
Following intravenous administration, the drug achieves 100% bioavailability. The distribution phase is rapid, with plasma concentrations peaking within 5 minutes. [7]
Oral Administration
Oral administration of methocarbamol is off-label in horses. Veterinarians determine an appropriate dosage based on the horse’s condition, body weight, pre-existing health conditions, and response to therapy.
The oral route is employed for less acute conditions, maintenance therapy, or ongoing management of chronic muscular pain. Oral administration is achieved using crushed human/canine tablets dispersed in water and delivered via nasogastric intubation or an oral dose syringe, or by utilizing flavored compounded powders, pastes, and suspensions.
The oral administration of methocarbamol results in significantly reduced bioavailability in horses compared with the intravenous form. [2][5][7]
This reduction is only seen in horses. It is attributed to incomplete gastrointestinal absorption within the equine hindgut combined with a high degree of liver metabolism during digestion of the drug.
Approximately 40% of the oral dose is extracted and metabolized by the equine liver before reaching systemic circulation. [5]
Because of this extensive “first-pass” metabolism, oral formulations must be administered at significantly higher concentrations than the intravenous equivalent to achieve comparable therapeutic plasma concentrations in horses. Further, because the drug is cleared rapidly, multi-dose daily administration intervals are frequently prescribed by veterinarians to maintain a consistent therapeutic effect.
Storage
Injectable solutions of methocarbamol are stored at room temperature, typically between 20 to 25°C (68 to 77°F). The formulation should be protected from excessive heat and direct light.
If the injectable solution is mixed with compatible IV infusion fluids by a veterinarian, the resulting mixture must not be refrigerated, as cooling may cause the methocarbamol to precipitate out of solution. [4]
Tablets, compounded powders, and suspensions must be stored at 15 to 30°C (59 to 86°F) in tightly sealed containers to protect them from moisture and ambient light. All formulations must be kept strictly out of reach of children and domestic animals. [4]
Safety Warnings & Precautions
The administration of methocarbamol requires strict adherence to safety protocols to prevent complications.
Key safety concerns include: [4]
- Vascular extravasation: If the drug is injected outside of the vein, it can cause severe local tissue necrosis (death), extreme pain, or immediate, life-threatening seizures.
- Syringe aspiration: During IV administration, blood aspirated into the syringe will not mix uniformly with the hypertonic methocarbamol solution. The veterinarian may safely inject the mixture or halt the injection when the plunger reaches the separated blood.
- Performance interference: Due to the drug’s inherent sedative properties and its potential to cause mild ataxia, horses should not be ridden, driven, or otherwise worked during methocarbamol use.
Monitoring
During and immediately following administration of methocarbamol, the horse should be closely monitored by veterinary personnel or the caretaker.
In particular, horses receiving methocarbamol are monitored for: [4][5][8]
- Central nervous system status: Continuous observation for excessive sedation, severe ataxia, or profound weakness is required, particularly during the peak plasma concentration window (15 to 60 minutes post-administration).
- Kidney function: In horses receiving prolonged therapy or high doses of the injectable formulation, renal parameters (BUN, creatinine) and urine output should be monitored due to the risks associated with the polyethylene glycol 300 co-solvent that is commonly used to prepare IV formulations.
Contraindications
Contraindications refer to circumstances where a drug is to be avoided or only used with caution.
The administration of methocarbamol is contraindicated in the following clinical scenarios: [2][4]
- Renal Pathology: The injectable formulation must not be administered to equine patients with known or suspected kidney disease. This strict contraindication is due to the vehicle used in the injection, polyethylene glycol 300 (PEG-300). In cases of compromised renal function, exposure to high loads of PEG-300 can worsen pre-existing acidosis and significantly increase urea retention, potentially leading to acute renal failure.
- Hypersensitivity: The drug is absolutely contraindicated in horses demonstrating known hypersensitivity or allergic reactions to methocarbamol or any of the inactive excipients within the formulation.
- Pregnancy and lactation: Comprehensive safety studies have not been conducted in pregnant or lactating mares. Methocarbamol should not be used during pregnancy unless the attending veterinarian determines that the potential clinical benefits significantly outweigh the unknown developmental hazards.
- Neurological disease: The drug should be used cautiously in horses with neurological deficits, even if mild, as it can worsen incoordination and weakness. Horses with profound ataxia can be a danger to themselves and handlers.
Methocarbamol must not be used in horses intended for food. [4]
Side Effects
Methocarbamol is generally considered safe and well-tolerated in horses when administered under veterinary guidance. Like all medications, methocarbamol carries some risk of side effects. The majority of these side effects are direct extensions of the drug’s primary action as a central nervous system depressant. [3][8]
Side effects of methocarbamol may include: [2][4][6][8]
- Lethargy: The most frequently observed side effect is mild to moderate sedation or lethargy. Horses may exhibit a loss of interest in their surroundings, decreased spontaneous locomotor activity, a lowered head carriage, drooping eyelids, and a characteristic “sawhorse” stance. Partial protrusion of the penis may be observed in geldings and stallions.
- Decreased motor function: At higher doses, depression of the spinal interneurons can lead to ataxia (incoordination), weakness, and stumbling.
- Ptyalism: Salivation or excessive drooling may occur.
- Appetite changes: Vomiting is a noted side effect in small animals; gastrointestinal upset in horses may manifest as inappetence.
- Discolored urine: The metabolism and renal excretion of methocarbamol can cause a harmless, temporary discoloration of the urine, rendering it dark, brown, black, or blue-green.
- Local injection site reactions: If the hypertonic injectable solution is inadvertently administered extravascularly (outside the vein), it causes severe local tissue irritation, swelling, phlebitis (vein inflammation), and potential tissue necrosis.
Acute Toxicity (Overdose)
Acute methocarbamol toxicity is a medical emergency resulting from profound over-depression of the central nervous system. Because the drug has a relatively high therapeutic index, massive doses are generally required to induce toxicosis, but individual sensitivities and concurrent renal or hepatic impairment can lower the toxic threshold.
Clinical signs of acute toxicity in horses include: [3]
- Extreme lethargy
- Profound ataxia leading to recumbency (inability to stand)
- Apneustic breathing (a distinct, abnormal pattern of breathing characterized by deep, gasping inhalation with a pause before exhaling)
- Nystagmus (involuntary eye movements)
- Severe hypotension (low blood pressure)
- Coma
In some instances, toxic levels suppress inhibitory pathways so severely that contradictory muscle rigidity, tremors, and seizures occur. [3]
Immediate veterinary intervention is required if a horse exhibits severe incoordination, collapses, begins experiencing seizures, displays abnormal breathing patterns, or if an accidental massive ingestion is suspected.
Drug Interactions
Methocarbamol’s status as a central nervous system depressant dictates its interaction profile with other pharmacological agents. Concurrent administration of methocarbamol with other drugs that modulate the CNS can lead to profound, potentially dangerous synergistic or additive effects. [2]
The following classes of medications represent significant drug interactions: [2][5][9]
- Alpha-2 adrenergic agonists: Drugs such as xylazine, detomidine, romifidine, and dexmedetomidine are potent sedatives used routinely in equine practice. Concurrent administration with methocarbamol will exponentially amplify CNS depression, potentially leading to severe ataxia, hypotension, and sudden recumbency.
- Phenothiazines: Tranquilizers such as acepromazine exert their effects via dopamine antagonism and alpha-1 adrenergic blockade. When combined with methocarbamol, the risk of profound sedation and extreme vasodilation (severe hypotension) is increased.
- NSAIDs: Phenylbutazone (“Bute”) is frequently prescribed with methocarbamol for muscle soreness. While generally safe under veterinary supervision, the interaction may slow down how quickly the horse’s body clears methocarbamol, which is critical for competition withdrawal times. Administration of NSAIDs with methocarbamol can lead to a longer withdrawal time before competition. Consult your veterinarian to determine best timing in competitive horses.
- Opioids: Analgesics such as butorphanol, morphine, and buprenorphine can interact with methocarbamol, leading to unpredictable neurological responses, ranging from increased respiratory depression to paradoxical CNS excitement.
- General anesthetics: Methocarbamol is frequently used as an adjunct to anesthesia. However, they reduce the required doses of induction agents (like ketamine or thiopental). Without appropriate downward dose adjustments of the primary anesthetic agents, severe cardiorespiratory depression can occur.
- Anticholinergics: Drugs such as atropine, glycopyrrolate or Buscopan/N-butylscopolammonium bromide should be used cautiously with methocarbamol due to overlapping systemic effects.
Methocarbamol is not compatible for administration with lactated Ringer’s solution. [2]
Regulatory Status & Legal Considerations
Methocarbamol is a prescription-only (Rx) medication used in horses to manage muscle spasms and related conditions. Federal law restricts this drug to use by or on the lawful order of a licensed veterinarian.
Veterinarians may prescribe methocarbamol in oral or injectable forms depending on the horse’s condition and clinical needs. As with all prescription medications, it must be administered under veterinary supervision to ensure appropriate dosing, monitoring, and treatment outcomes.
Veterinarians may use oral formulations of methocarbamol for horses, which differs from the approved use on the product label. This type of use is permitted under established veterinary prescribing regulations and relies on professional judgment, available research, and clinical experience with the drug in horses.
Because methocarbamol affects the central nervous system and can alter muscle tone, coordination, and movement, its use is regulated in many equestrian competitions.
Equine sports governing bodies may impose withdrawal times, detection thresholds, and other restrictions to ensure fair competition and protect horse welfare.
If your veterinarian recommends methocarbamol, it is appropriate to discuss the expected benefits, duration of therapy, potential side effects, and any competition withdrawal requirements that may apply.

Status in Competition
The status of methocarbamol varies significantly depending on the governing body sanctioning the competition.
Current guidelines are as follows: [7][10][11][12][13]
- USEF: Restricted Therapeutic. Strict maximum 24-hour dose limits apply. Must not be administered within 12 hours prior to competition.
- RMTC/ARCI: Class 3 drug. Required withdrawal time, 48 – 72 hours (IV single dose). 1.0 ng/mL plasma threshold. Oral doses and repeated dosing require significantly longer withdrawal times due to bioaccumulation. Additionally, because oral methocarbamol is uniquely metabolized into guaifenesin (which is itself a regulated substance), metabolic accumulation warrants extended withdrawal intervals to avoid dual-substance violations.
- Equestrian Canada: Conditional. Required withdrawal time, 24 hours (single dose), 48 hours (5-day course). Elimination guidelines apply to both IV and oral routes.
- FEI: Controlled Medication. Required withdrawal time: 48 hours (Detection Time). Must be cleared prior to event. Relies on laboratory detection limits; requires consultation with the treating veterinarian to determine safe withdrawal periods based on the horse’s metabolism.
Always check the most current rules for your sport before starting your horse on a new medication.
Frequently Asked Questions
Here are some frequently asked questions about methocarbamol use in horses:
Methocarbamol is a central nervous system muscle relaxant commonly used to treat skeletal muscle spasms in horses. Veterinarians often prescribe it for conditions such as rhabdomyolysis (tying-up), muscle trauma, and certain neurological conditions that cause muscle rigidity. It may also be used as supportive therapy in horses with tetanus.
Methocarbamol works by depressing nerve activity within the central nervous system. Instead of acting directly on muscle tissue, it reduces abnormal nerve signals from the brain and spinal cord that trigger excessive muscle contractions, helping relieve spasms and stiffness.
Methocarbamol may be administered intravenously by a veterinarian for rapid effect in acute cases. For ongoing treatment, it is commonly given orally as tablets or powder mixed with feed. The appropriate dose and duration of treatment should always be determined by a veterinarian.
When administered intravenously, methocarbamol may begin to reduce muscle spasms within minutes. Oral formulations typically take longer to take effect, with improvement often observed within a few hours depending on the horse and the underlying condition.
The duration of action varies depending on the dose and route of administration, but its muscle-relaxing effects generally last several hours. Veterinarians may recommend multiple doses per day when treating ongoing muscle spasms.
Methocarbamol is generally well-tolerated, but some horses may develop mild sedation, lethargy, or incoordination (ataxia). Less commonly, sweating or increased salivation may occur. Any unusual reactions should be reported to a veterinarian.
Because methocarbamol depresses the central nervous system, it may cause mild sedation or reduced coordination. Horses receiving this medication are usually not suitable for athletic work until the drug has worn off and the underlying condition has resolved.
Methocarbamol and its metabolites are eliminated through the kidneys and can temporarily change the color of a horse's urine. Dark brown, black, or blue-green urine may occur and is usually harmless, resolving once the medication is cleared from the body.
Methocarbamol is considered a regulated medication in many equestrian disciplines. Withdrawal times vary depending on the governing organization and dose used, so riders and trainers should consult competition rules and their veterinarian before competing.
Methocarbamol is often used alongside other treatments, such as anti-inflammatory drugs, fluid therapy, or supportive care, depending on the underlying condition. However, all medication combinations should be approved by a veterinarian to avoid potential interactions.
Alternative treatments depend on the cause of the muscle spasm. Management may include rest, hydration, anti-inflammatory medications, electrolyte or mineral support, or other muscle relaxants. A veterinarian can determine the most appropriate treatment plan.
Summary
Methocarbamol is a centrally acting skeletal muscle relaxant used in horses to reduce painful muscle spasms caused by injury, inflammation, or neurological disease. It works by decreasing abnormal nerve signals in the brain and spinal cord rather than acting directly on muscle tissue.
- Methocarbamol is commonly used to treat muscle spasms associated with rhabdomyolysis (tying-up), trauma, inflammatory muscle conditions, and tetanus
- The drug reduces muscle hyperactivity by depressing reflex pathways in the central nervous system while generally preserving voluntary muscle movement
- Intravenous methocarbamol is FDA-approved for horses, while oral tablets and compounded formulations are used off-label for ongoing management
- Common side effects include sedation, lethargy, and incoordination due to its central nervous system depressant effects
- Because methocarbamol can affect coordination and performance, its use is regulated in many equestrian competitions and requires appropriate withdrawal times
- Methocarbamol is a prescription medication that must be administered under veterinary supervision to ensure safe dosing and monitoring
References
- Methocarbamol. PubChem.
- Plumb's Veterinary Drugs. John Wiley & Sons. 2018.
- Dowling. P. M. Skeletal Muscle Relaxants for Animals. Merck Veterinary Manual. 2025.
- Robaxin Product Insert. Zoetis. 2003.
- Cunningham. F. E. et al. The Pharmacokinetics of Methocarbamol in the Thoroughbred Race Horse. Journal of Veterinary Pharmacology and Therapeutics. 1992.
- Methocarbamol (Robaxin®). Pet MD. 2022.
- RMTC CTS Monograph Series: Methocarbamol. RMTC. 2020.
- Muir. W. W. et al. The Pharmacology and Pharmacokinetics of High‐dose Methocarbamol in Horses. Equine Veterinary Journal. 1992.
- Cynthia Cole et al. Equine Pharmacology. 2015.
- 2026 USEF Guidelines & Rules for Drugs and Medications. USEF. 2025.
- RMTC Withdrawal Guidance Advisory. RMTC. 2023.
- Equestrian Canada: Equine Medication Control Guide. Equestrian Canada. 2025.
- FEI Clean Sport. FEI. 2025.